Purpose and Objective
The purpose of this project is to implement smartphone technology to collect behavioural data related to risk perceptions, knowledge and personal protective practices during the COVID-19 outbreak. Our objective is to collect actionable data that will be used to inform public health strategies and predictive models.
The expected length of the study is approximately four months with an estimated weekly time commitment of about 15 to 20 minutes. Your participation will provide vital data to help understand the context behind the decisions that individuals make during the COVID-19 outbreak.
Study Procedures
Smartphone data gathering will begin after downloading the Ethica app (free from the app store) and providing consent.
Data will be gathered from all participants in the form of:
Geographical Data
Ethica will collect location data that will only be used to study movement trends in the population. As long as the smartphone is in discoverable mode, location data will be collected via the smartphone’s Global Positioning System (GPS). At any time, participants can suspend data collection by simply clicking on the pause button. No location information will be used for any other purpose. Location information will only be published in aggregate to remove the chance of identifying persons. If participants choose to opt out of GPS data collection, they will be given an alternative option to instead provide the first three digits of their postal code.
App Triggered Surveys:
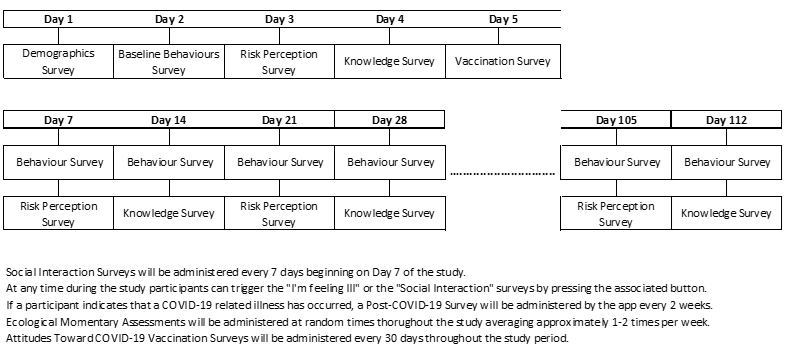
Mini-surveys will be conducted throughout the study period. In Phase 1, one mini-survey will be administered per day for the first 5 days of the study to collect baseline data on risk perceptions, behaviours and knowledge pertaining to COVID-19. In Phase 2, two short mini-surveys will be administered on a weekly basis for the remainder of the study period (approximately four months) to assess risk perceptions, behaviours, knowledge, attitudes toward vaccination, occurrence of illness and social contacts on an ongoing basis. A survey called the Ecological Momentary Assessment Survey will also be administered approximately 1-2 times per week to gather information on activities and surroundings during times when the surveys are completed. Short weekly surveys about social interactions and monthly surveys about attitudes to vaccines will also be administered (see the Timeline below for details).
User Triggered Surveys:
There will be a button for participants to trigger surveys related to experiencing illness and participating in social gatherings.
Focus groups:
Participants will have the option of providing further qualitative data by participating in virtual focus groups to provide insights into their risk perception and management, attitudes and beliefs, and personal protective behaviour as it relates to the COVID-19 pandemic. Participants who opt in for focus groups will be asked to sign a subsequent consent form at a later date.
Timeline