Western Canada
Regional prevalence data for gastrointestinal parasites, heartworm, fleas, and ticks for the provinces in Western Canada.
Region-specific parasites
- Ixodes pacificus (Western black-legged tick)
- Pulex simulans (wildlife flea) more common than Ctenocephalides felis (cat flea)
- Echinococcus multilocularis
Gastrointestinal parasites
Data for this section are drawn from the recent national fecal survey of shelter dogs (n=1086) and cats (n=636) in 26 shelters across Canada (Villeneuve et al., 2015), and recently published findings in owned, shelter, and free ranging population of dogs and cats in western Canada. The tables below from the national survey show fecal flotation data from shelter animals, about half of which were less than a year old, and are therefore not representative of the expected parasite prevalence in client-owned pets that receive regular veterinary care. However, they do provide information on the types of parasites that pets may be exposed to in this region, as most of these animals are locally sourced and have been free-ranging at some point.
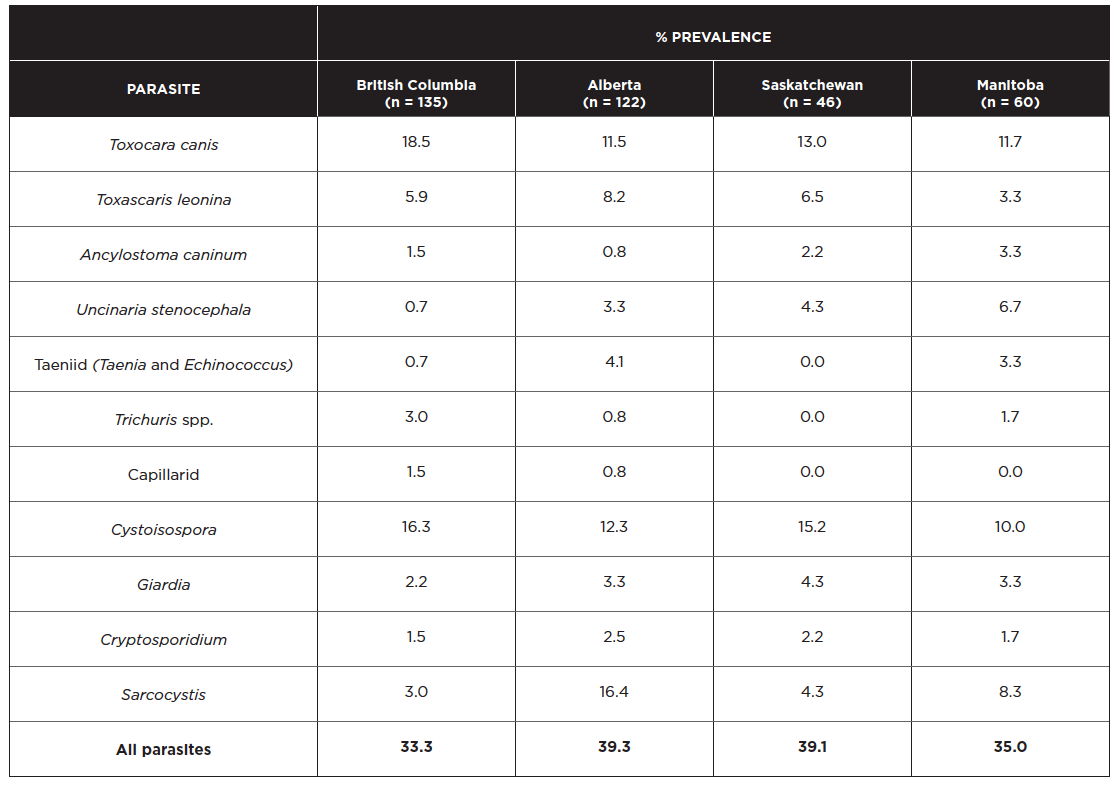
In centrifugal flotations on fecal samples from 363 dogs in shelters in BC, AB, SK, and MB, the most commonly observed parasites were Toxocara canis (14%), Toxascaris leonina (6%), and Cystoisospora (14%) (Villeneuve et al 2015). In general, prevalence of Toxascaris was higher in western Canada (6-7%) than nationally (3%). The province-specific results are shown below, with the total number of dogs sampled per province in parentheses.
Comparison of fecal flotation results from shelter, rural, and remote dogs from Saskatchewan to pet dogs in Saskatoon showed that similar types of parasites were found, though prevalence in pets was much lower (Gaunt and Carr 2011, Schurer et al 2012, Schurer et al 2013, Schurer et al 2014). Similarly, for pet dogs in Calgary, Alberta, ascarid (1.9%) and hookworm (0.8%) infections were much less prevalent than in shelter dogs, while Giardia (9.2%) was more so (Joffe et al 2011).
Other parasites present at low frequency in dogs in western Canada include flukes such as Alaria spp. and Metorchis conjunctus, the cestode Diphyllobothrium, and the giant kidney worm Dioctophyme renale (Gaunt and Carr 2011, Schurer et al 2012, Schurer et al 2014a, Schurer et al 2014b), all of which are acquired from ingestion of fish, frogs, or wildlife paratenic hosts.
In the national survey, dogs and cats in western Canada had higher fecal prevalence of taeniid cestode eggs (3.1% and 6.3%) than the national population (1.6% and 4.4%), possibly due to increased access to wildlife intermediate hosts. While most of these eggs are likely Taenia pisiformis (in dogs) and T. taeniaeformis (in cats), they could also be Echinococcus multilocularis or the cervid strains of E. granulosus (E. canadensis), as the eggs are indistinguishable from each other and from the eggs of Taenia species. Both species of Echinococcus are zoonotic, in contrast with Taenia spp. Molecular characterization detected DNA of E. canadensis, but not E. multilocularis, in the feces of 4 stray dogs from British Columbia, Alberta, and Ontario (Villeneuve et al 2015) and in 6% of 155 fecal samples from dogs in a community in Saskatchewan with a human case (Himsworth et al 2010). E. canadensis is endemic in most of Canada, causes cystic echinococcosis in people, and dogs can serve as definitive hosts and a source of human exposure. Therefore, prophylactic treatment of dogs with access to cervid carcasses is indicated wherever wildlife reservoirs exist (all of Canada except the High Arctic Islands and Atlantic regions) (Deplazes et al 2017). E. multilocularis is established at high prevalence in wild canids across northwestern Canada, including coyotes, foxes, and wolves (Jenkins et al 2013, Deplazes et al 2017). Dogs can also serve as definitive hosts for this parasite, carrying the adult cestodes in their intestine, generally without any ill effects. However, canine alveolar echinococcosis (AE) due to E. multilocularis – with dogs acting as the intermediate host rather than the definitive host – has been diagnosed in all 4 western provinces in recent years (Peregrine 2015, Villeneuve et al 2015, Deplazes et al 2017). AE (in dogs and people) generally develops as a “parasitic tumor” in the liver and can be fatal if not detected early and treated aggressively. Canine alveolar echinococcosis appears to be emerging in this region, possibly as a result of introduced European strains of the parasite in addition to North American strains long established in the prairie regions of western Canada (Jenkins et al 2013, Deplazes et al 2017). In order to mitigate the danger of alveolar echinococcosis for people, dogs should not be allowed access to rodent intermediate hosts, owners should avoid contact with food and water potentially contaminated with feces of wild canids, and high-risk dogs (those with access to wild rodents) should be treated monthly with praziquantel, year-round. Protecting dogs from alveolar echinococcosis involves prevention of contact with habitats, food, and water contaminated with feces of wild canids, even in urban regions such as dog parks and golf courses.
Common protozoan parasites seen in dogs in western Canada include Cystoisospora and Sarcocystis, which are not zoonotic, as well as Giardia and Cryptosporidium, which are both possibly zoonotic depending on the species/genotype. Dogs in rural and remote regions of Western Canada are generally infected with the zoonotic genotypes of Giardia, in contrast to kennel dogs in central Canada that are infected with the dog-specific genotypes (Jenkins et al 2013). In the national fecal survey of shelter dogs, dogs in western Canada had more than double the prevalence of Sarcocystis (12%) than the national population (5%) (Villeneuve et al 2015), likely due to higher access to wildlife intermediate hosts and/or raw food diets.
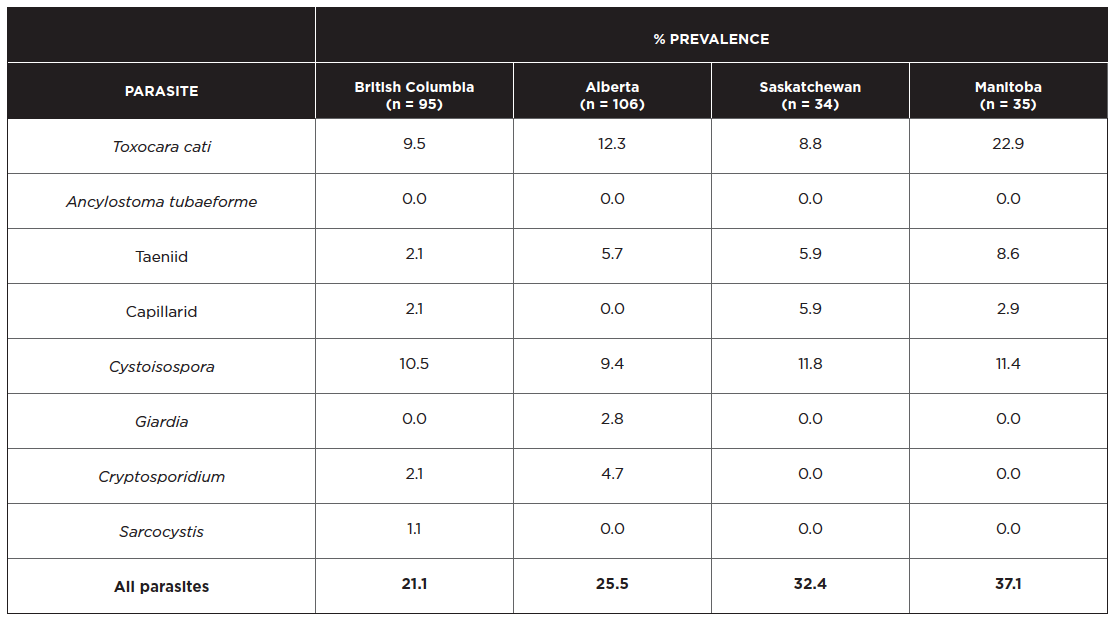
Based on centrifugal flotations of fecal samples collected from 270 shelter cats in Western Canada (BC, AB, SK and MB), the most commonly observed helminth parasites in cats are Toxocara cati (12%), taeniid tapeworms (5%), and Cystoisospora (10%) (Villeneuve et al 2015). The province-specific data are shown below, with the total number of cats sampled per province in parentheses.
Although not detected in the national survey, cats can also acquire Toxascaris leonina and, rarely, Echinococcus multilocularis. The latter has been found in cats in Saskatoon (Wobeser 1971), but cats are not thought to be epidemiologically significant in transmission.
Examination of 635 cat fecal samples submitted to the Saskatchewan provincial veterinary diagnostic laboratory also found that Toxocara cati (4.7%), Cystoisospora (3.8%), and taeniid tapeworms (1.3%) are the most common intestinal parasites in cats in western Canada (Hoopes et al 2013). The lower overall prevalences as compared to the shelter cats are expected since pet cats are more likely to receive veterinary care and less likely to be exposed through diet and water. In the same study, Giardia (9.9%) and Cryptosporidium (2.3%) were found to be much more prevalent than suggested by fecal flotation data due to the greater sensitivity of the immunofluorescent detection method used. Finally, as for dogs in western Canada, endoparasite prevalence and diversity was higher in samples from rural (41%) and free-roaming (32%) than client-owned cats (6%) in SK and AB (Hoopes et al 2015).
Heartworm
Southern Manitoba and the southern Okanagan Valley in BC are the only regions in Western Canada currently considered endemic for heartworm. In these regions, regular testing should occur in April-May and monthly preventives should be administered from June 1st to November 1st. The last systematic postal survey of heartworm in Canada was carried out in 2010. Of the 564 canine cases diagnosed, 77 (14%) were in Manitoba, 8 (1%) were in British Columbia, and 2 (0.35%) were reported in each of Alberta and Saskatchewan. All positive dogs in AB and SK had a history of travel or were imported from other countries.
The 2010 postal survey data for Western Canada are presented below alongside the postal survey from 2002 and IDEXX SNAP 4Dx data from 2013-2014 (Herrin et al 2017). Because the 2013-2014 SNAP 4Dx test results are not accompanied by travel history or information on confirmatory testing, we cannot draw firm conclusions when comparing this data to veterinarian- diagnosed cases from prior years.
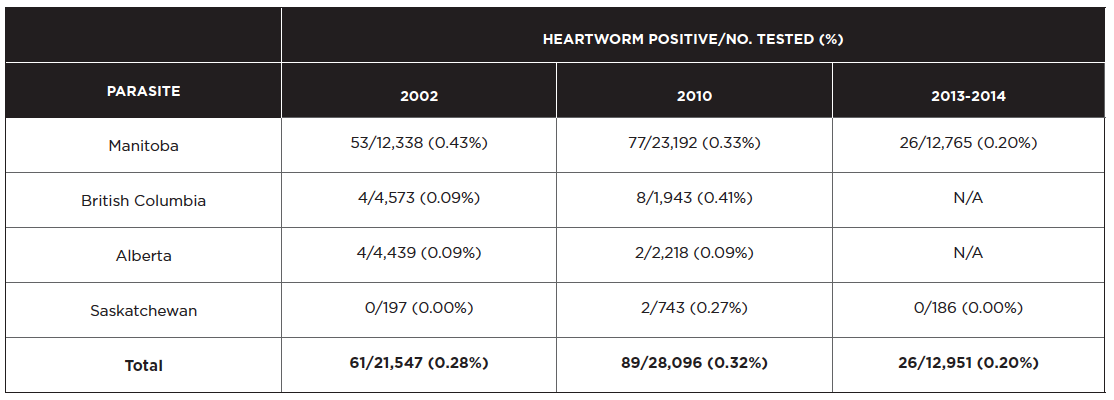
Overall, the prevalence of heartworm infection in Western Canada appears to be very low (0 to <1%), generally associated with living in or travel to an endemic region, and relatively unchanged over time. Veterinarians in the southern Okanagan Valley and southern Manitoba need to remain vigilant in their testing and continue to recommend heartworm prevention for their patients, and all veterinarians need to take a good travel history.
Fleas
There are no prevalence data for fleas in western Canada.
Anecdotally, dogs and cats in the lower mainland of British Columbia are at risk almost year-round for infestations with Ctenocephalides felis and/or Ctenocephalides canis, and flea prevention should be prescribed accordingly.
On the prairies and in eastern British Columbia, the prevalence of flea infestation of pets and households is quite low and is not usually associated with Ctenocephalides felis (cat flea), which is the most common flea of dogs and cats in many parts of the world. Infestations in this area of western Canada are usually associated with Pulex simulans from infested wildlife species (e.g. foxes, coyotes, skunks, birds) that have established themselves in a neighborhood, or to areas frequented by such wildlife.
Ticks
Dermacentor variabilis and D. andersoni are the most commonly found ticks on dogs and cats in western Canada, generally as adult ticks in May-June, although this season is likely to increase as climate continues to warm. Dermacentor andersoni is present in BC, AB, and western SK, while D. variabilis is present in MB, SK, and eastern AB. Published data demonstrate that the distribution of D. variabilis is expanding west into western Saskatchewan and Alberta, and both D. variabilis and D. andersoni are moving north (Dergousoff et al 2013). Rarely, wildlife-origin ticks such as Dermacentor albipictus (moose tick), Ixodes kingi, and Haemaphysalis leporispalustris (rabbit tick) may be found on pets in western Canada, and Rhipicephalus sanguineus is occasionally found on dogs with a history of travel outside western Canada.
Ixodes scapularis is established in southern Manitoba, and I. pacificus in coastal British Columbia. Outside of these regions, I. scapularis that are periodically found on dogs (generally in fall) have likely been acquired from migratory birds. Overall, more than 15% of I. scapularis are infected with the Lyme disease agent, Borrelia burgdorferi, while less than 10% of I. pacificus are infected (Bouchard et al 2015).
» Maps of Lyme disease risk areas are generated by public health agencies and are available through the links below.
British Columbia: www.bccdc.ca/health-info/diseases-conditions/lyme-disease-borrelia-burgdorferi-infection
Manitoba: www.gov.mb.ca/health/publichealth/cdc/tickborne/surveillance.html
In Western Canada, the incidence of tick-borne disease is considered to be low. Of 240 dogs tested in Saskatchewan and Alberta, 2.5% had antibodies to the organism that causes Rocky Mountain spotted fever (Leighton et al 1995). More recently there have been reports of Anaplasma phagocytophilum in British Columbia dogs (Lester et al 2005, Kowalski et al 2015), and there are periodic reports of A. phagocytophilum in Saskatoon dogs (Cockwill et al 2009). One study using the IDEXX 4Dx SNAP test found that 3% of 77 dogs in the southeastern portion of Saskatchewan were positive for antibodies to B. burgdorferi, likely due to spillover from the endemic region in southern Manitoba (Schurer et al 2014b). This may also support the western spread of I. scapularis, as projected by models based on climate change projections.
Data from a total of 115,636 Canadian dogs tested in 2013-2014 (Herrin et al 2017), compared to 94,928 samples in 2007 (from Incidence of Heartworm, Ehrlichia canis, Lyme Disease, Anaplasmosis in dogs across Canada as determined by the IDEXX SNAP® 3Dx® and 4Dx® Tests – 2007 National Incidence Study Results, IDEXX, Markham, ON) indicate seroprevalence for tick-borne agents was as follows:
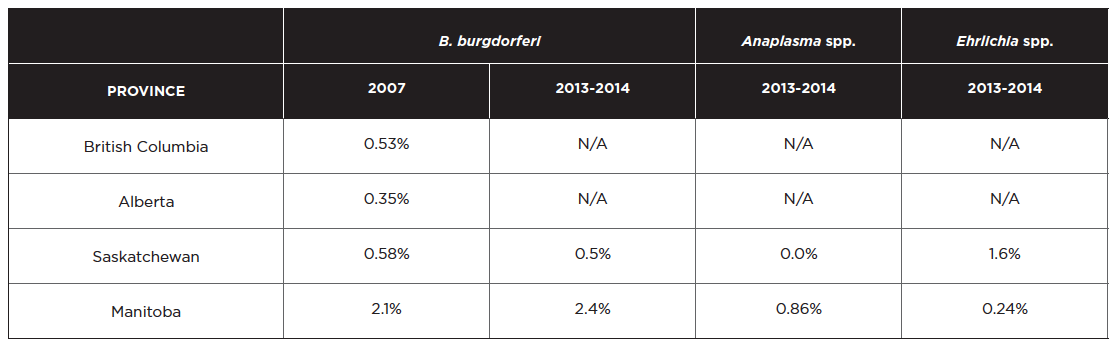
These data should be interpreted carefully as they indicate exposure and not current infection. Many dogs that seroconvert for B. burgdorferi and other vector borne diseases do not develop clinical signs. The true prevalence of infection may be lower since there is no evidence that confirmatory tests were done for any of the samples. It is also not known if exposure occurred locally, and, in light of the very low prevalence of infection in most areas, some positive results may be false positives.
